Pharmacy: Safe Online Buying, Compounding, and Practical Tips
Buying medicine online can save time and money, but it also brings risks. This page pulls together clear, practical guides and reviews so you know where to look, what to ask, and how to stay safe. You’ll find straightforward advice for patients and quick checklists for pharmacists—all in one place.
How to spot a safe online pharmacy
Start by checking for a real pharmacy address and phone number. If a site hides contact details or only offers chatbots, be cautious. Legit pharmacies will ask for a prescription for prescription-only drugs. If a site sells controlled meds without a prescription, it’s a red flag.
Look for third-party verification. In the US, the Verified Internet Pharmacy Practice Sites (VIPPS) seal helps. In the UK and Europe, regulators list licensed services—cross-check the pharmacy name on official regulator sites. Also, read recent customer reviews on independent platforms; focus on delivery accuracy and product quality.
Check the product pages. A reliable pharmacy lists active ingredients, dosage forms, batch numbers, and manufacturer details. If descriptions are vague or use stock photos only, pause. Pricing that’s dramatically lower than usual can hide counterfeit or expired meds.
Compounded meds, prescriptions, and pharmacist checks
Compounding means tailoring medication to a patient’s needs—different dose, flavor, or form. Compounded meds should be handled by licensed compounding pharmacies with clear quality and sterility practices. Ask for documentation about their procedures and how they test purity.
If you’re a patient, keep your prescriber in the loop when using compounded drugs. Ask your pharmacist which standards and testing methods they use. For pharmacists filling co-prescriptions, use a short screening checklist: verify current meds, flag interactions (especially with tricyclics, anticoagulants, or seizure drugs), confirm renal/hepatic status, and document counseling points.
Delivery and storage matter. Sensitive meds may need temperature control. Choose shipping options with tracking and, when needed, cold-chain handling. Store meds as the label directs and dispose of leftovers safely at take-back points.
On this category page you’ll find articles on buying specific drugs safely, reviews of online pharmacies, and practical pharmacist guides. Read post summaries, check dates, and use the tips to compare options fast. If something feels off, call your prescriber or local pharmacy—an expert opinion beats a risky purchase.
If you want, start with guides about buying antidepressants, methotrexate, or oxcarbazepine online, or read the pharmacist checklist for imipramine co-prescriptions. These posts walk through real steps and legal points so you can make safer choices today.

- 0 Comments
Explore the updated UK substitution laws for 2026, covering NHS policies on generic medicines, service shifts to community care, and the impact of 2025 regulatory reforms on patients and providers.

- 13 Comments
Learn how to check for pharmacy-level drug recall notifications using FDA and TGA systems, integrated software, and verification steps to protect patients and avoid legal risks.

- 13 Comments
Pharmacy dispensing errors are more common than you think-and they’re preventable. Learn the top mistakes, why they happen, and how pharmacists and patients can stop them before they hurt someone.
- 10 Comments
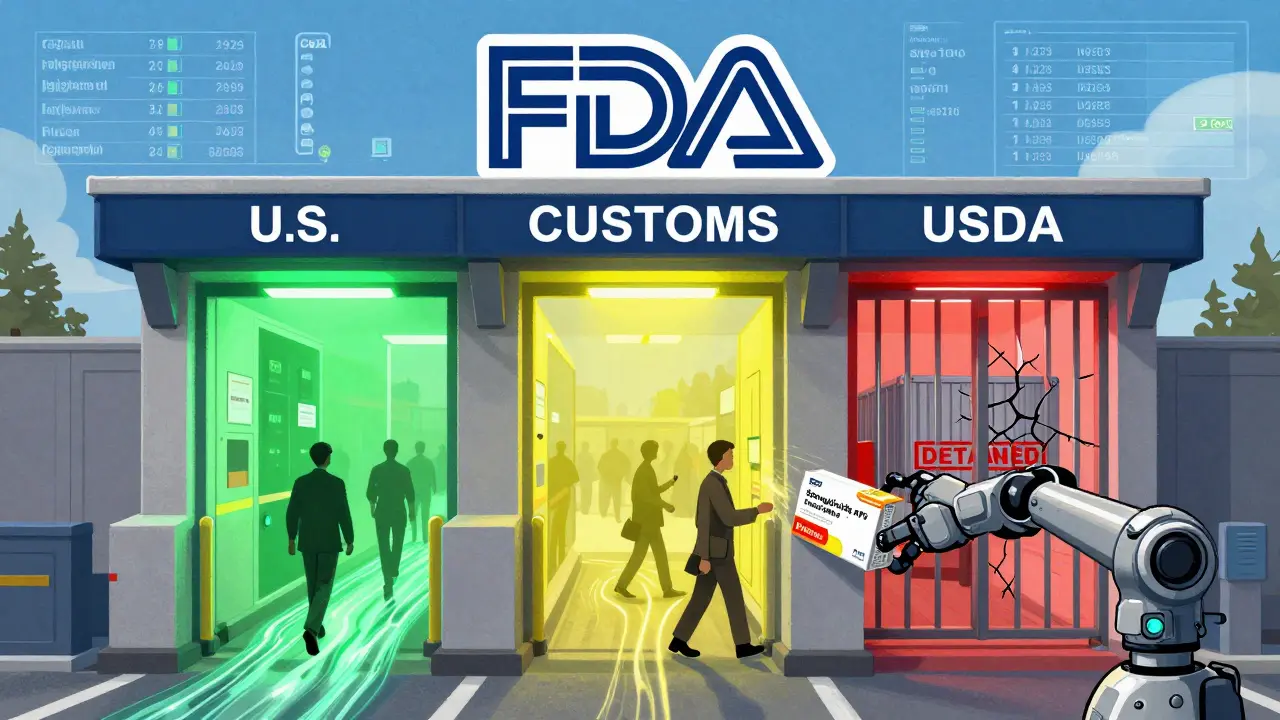
The FDA uses Import Alerts to block drugs from non-compliant manufacturers before they reach U.S. pharmacies. With the Green List system, only verified suppliers get through-others face automatic detention at the border.

- 11 Comments
Generic drugs must meet the same FDA standards as brand-name drugs, including pharmaceutical and bioequivalence. Learn how testing, manufacturing rules, and inspections ensure quality and safety.

- 14 Comments
Every part of your prescription label has a purpose - from your name to the expiration date. Understanding what each section means can prevent dangerous mistakes and help you take your medicine safely.

- 10 Comments
Authorized generics let brand drugmakers launch their own low-cost versions during generic exclusivity, undercutting independent competitors and slowing price drops. This legal but controversial tactic distorts competition and keeps drug prices higher than they should be.

- 16 Comments
Learn how to safely check supplement-drug interactions using verified databases like NatMed. Step-by-step guide for clinicians to prevent dangerous adverse events and improve patient safety.
- 8 Comments
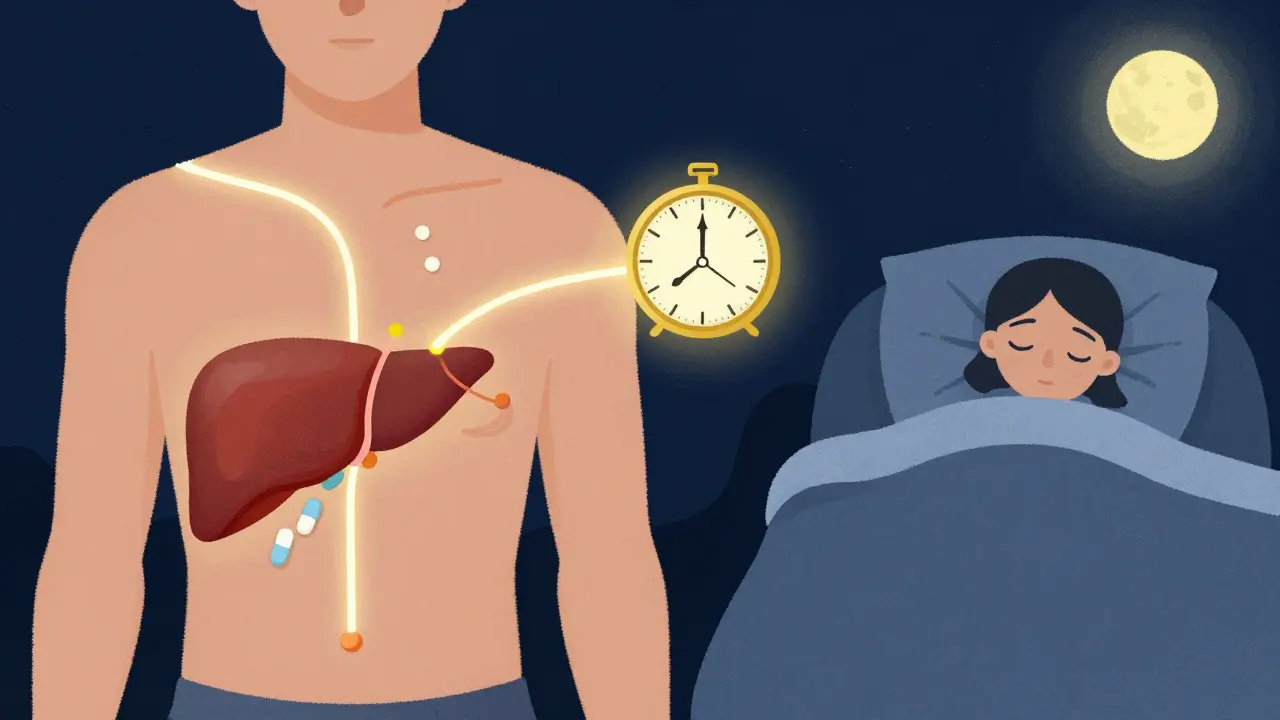
The time you take your medication can reduce side effects and boost effectiveness. Learn how chronotherapy uses your body’s natural rhythms to make drugs safer and more powerful.

- 9 Comments
The Hatch-Waxman Act of 1984 balanced drug innovation and generic access. It created the ANDA pathway, patent term restoration, and Paragraph IV challenges. Today, generics make up 90% of prescriptions but only 18% of spending. Learn how this law works and its ongoing challenges.
