Category: Pharmacy - Page 3
- 14 Comments
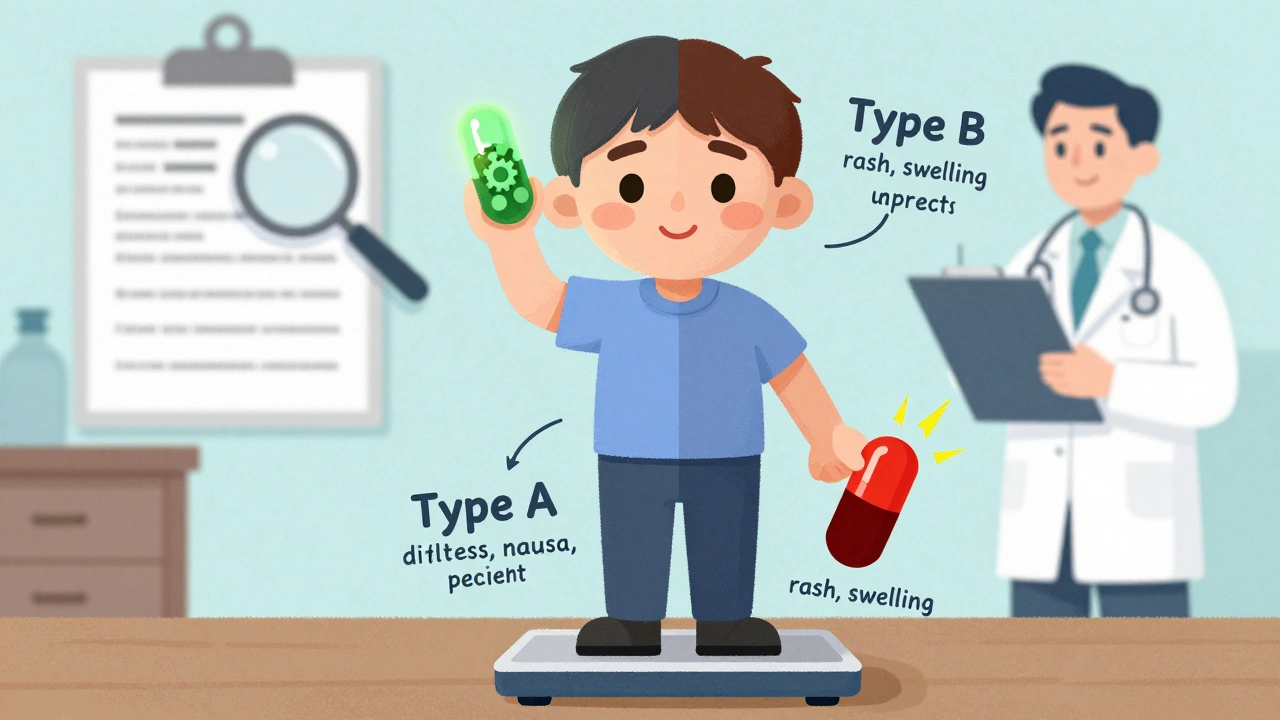
Learn the key differences between Type A and Type B adverse drug reactions - why some side effects are common and predictable, while others are rare but dangerous. Understand how to recognize, prevent, and report them.
- 11 Comments
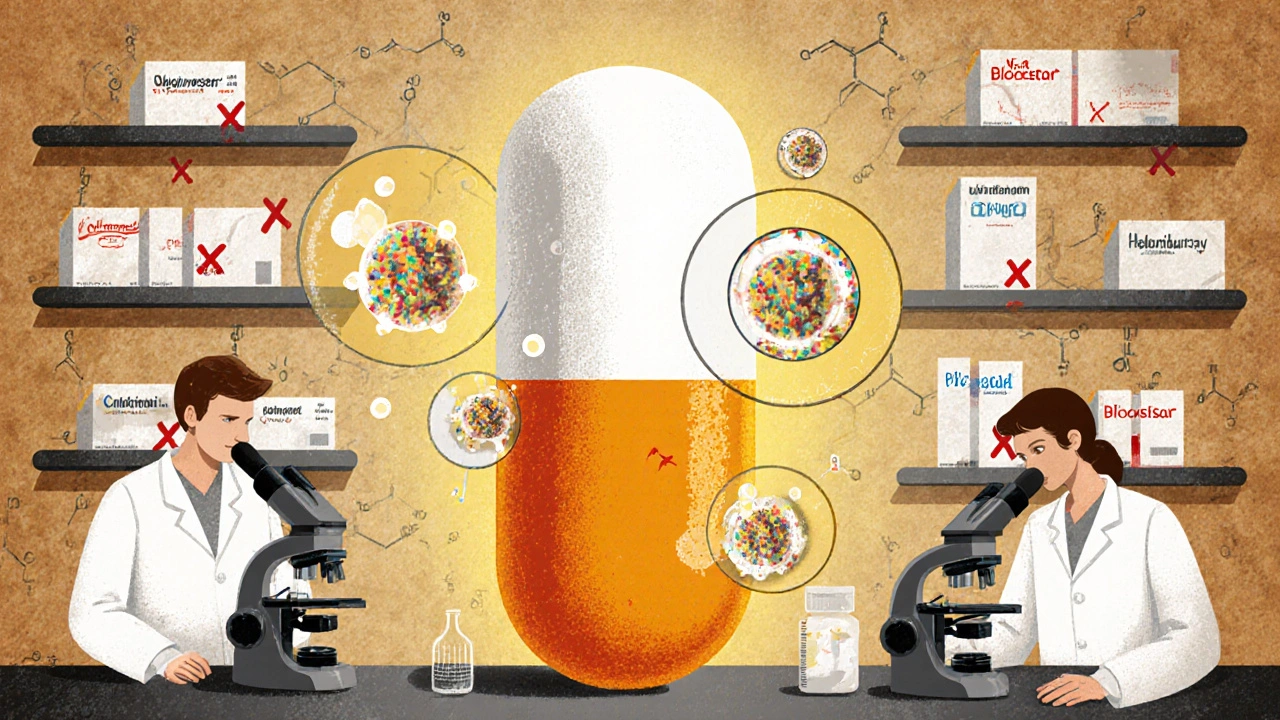
Generic drugs save billions in healthcare costs and make up 90% of U.S. prescriptions. Learn how they're made, from reverse-engineering brand-name pills to FDA-approved manufacturing under strict quality controls.
- 13 Comments
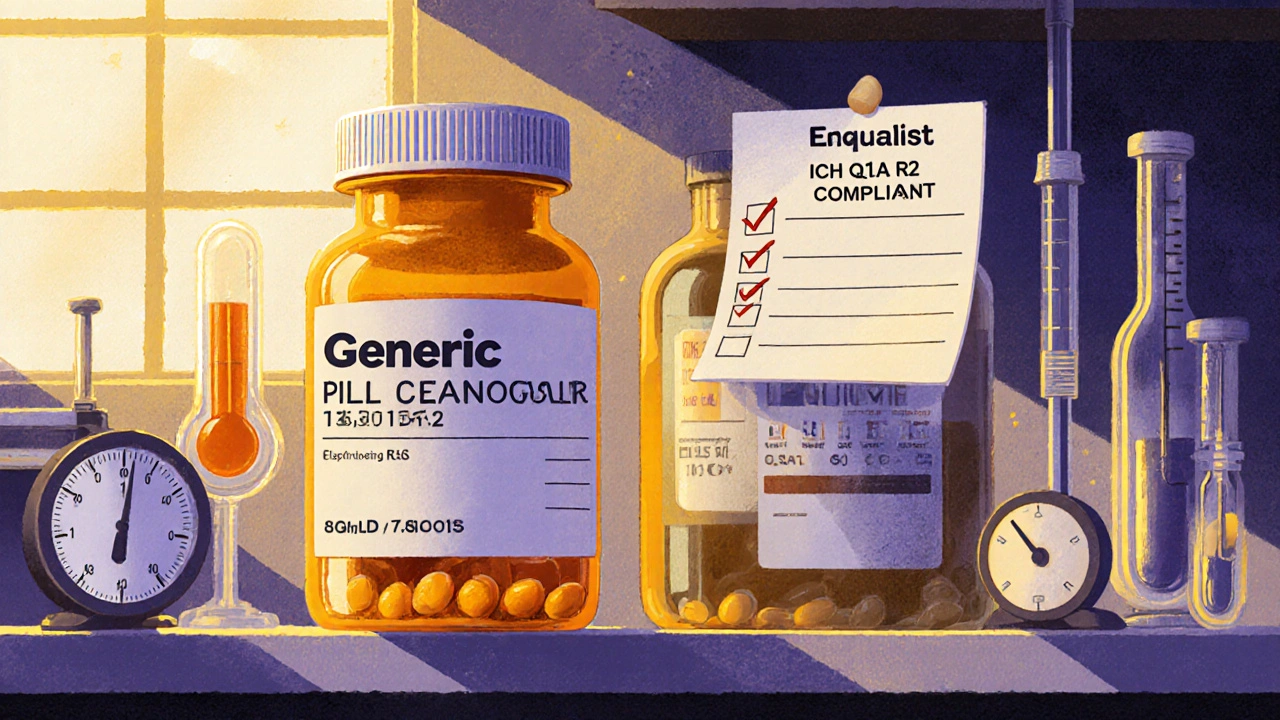
Learn exactly what the FDA requires for stability testing of generic drugs, from batch testing and storage conditions to common rejection reasons and upcoming 2025 changes that impact approval timelines.

- 10 Comments
Dulcolax (bisacodyl) works fast for constipation but isn't safe long-term. Compare it to stool softeners, magnesium, psyllium, and lactulose to find the right fit for your needs.

- 15 Comments
Compounded medications offer personalized treatment when standard drugs don't work-but they come with risks. Learn when they're necessary, how to find a safe pharmacy, and what to watch out for.

- 14 Comments
Learn how prior authorization for generic alternatives works in pharmacy practice, why insurers require it, how to navigate denials, and what changes are coming in 2026 to reduce delays and improve patient care.
- 8 Comments
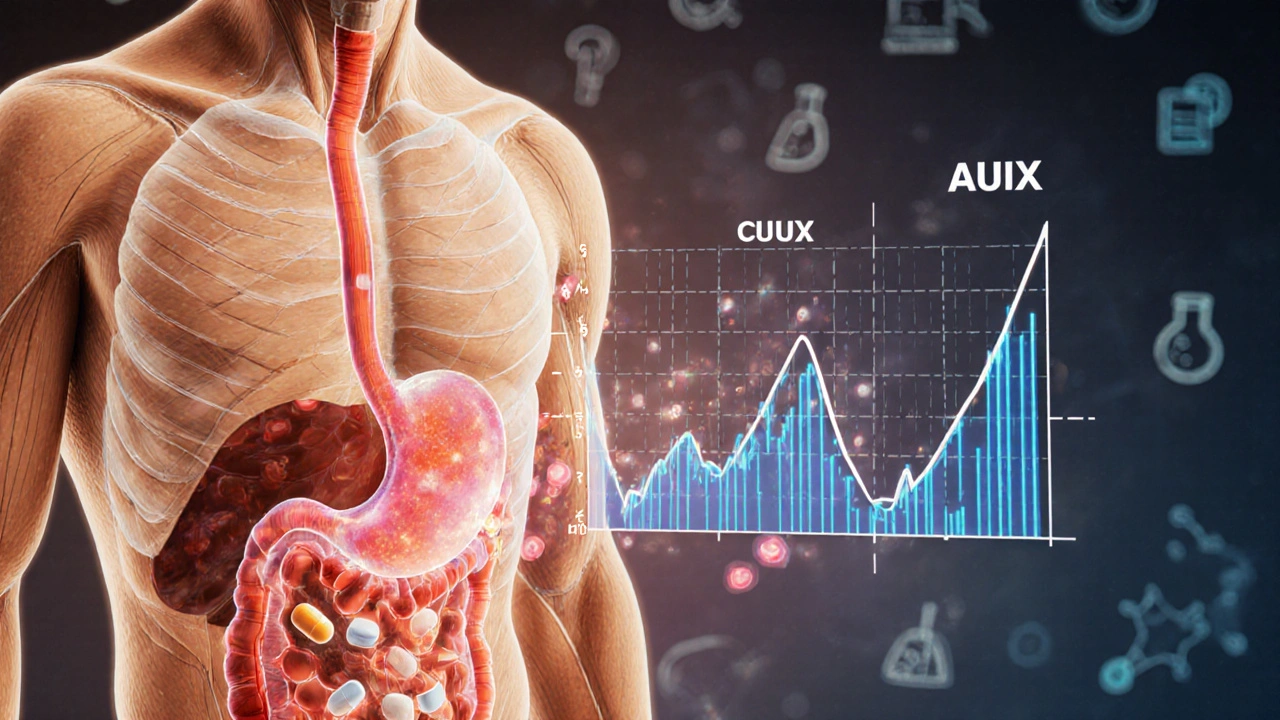
In vivo bioequivalence testing uses human trials to measure drug absorption, while in vitro methods rely on lab tests like dissolution. Learn when each is used for generic drug approval and why regulators are shifting toward in vitro for simpler products.
- 14 Comments
Discover how generic drugs go from FDA approval via ANDA to being available at your local pharmacy. Learn about the science, regulation, and supply chain that make affordable medications possible.

- 9 Comments
Learn how to protect your health data when using online pharmacies in 2025. Discover the .pharmacy domain, VIPPS seal, and key red flags that separate safe sites from dangerous ones.

- 9 Comments
Salbutamol is essential for asthma and COPD patients, but prices vary widely. Learn how to save money with generics, insurance tips, and affordable options in 2025.
